|
After adding electron, the same atom becomes a negative ion. 
In contrary to ionization energy, the electron affinity is defined as the change in the energy state, when an extra electron is introduced into an atom (when it is in the neutral state). Coming to periodic table trends regarding this, the first ionization energy goes higher from left to right direction, while it drops from top to bottom direction. After detaching the electron, the same atom turns into positive ion. The ionization energy of an element is defined as the total energy required to remove an electron from an atom (when it is in the neutral state). When a comparison is made between the two ionic forms of a same element, it is found that the positive charged ion has a smaller radius than the radius of the negatively charged ion. Similar to the atomic radius, the radii of positive ions (cations) and negative ions (anions) of chemical elements can be measured. The atomic radii of elements gradually increases as you move across (from right to left) in a period, and down (from top to bottom) in a group. Measure distance between the centers of the two adjacent atoms and half of the value is called atomic radius of the element. Atomic Radius TrendĬonsider two atoms of the same element that touch each other by their outer edges. Detailed information regarding trends in the modern periodic table is highlighted below. 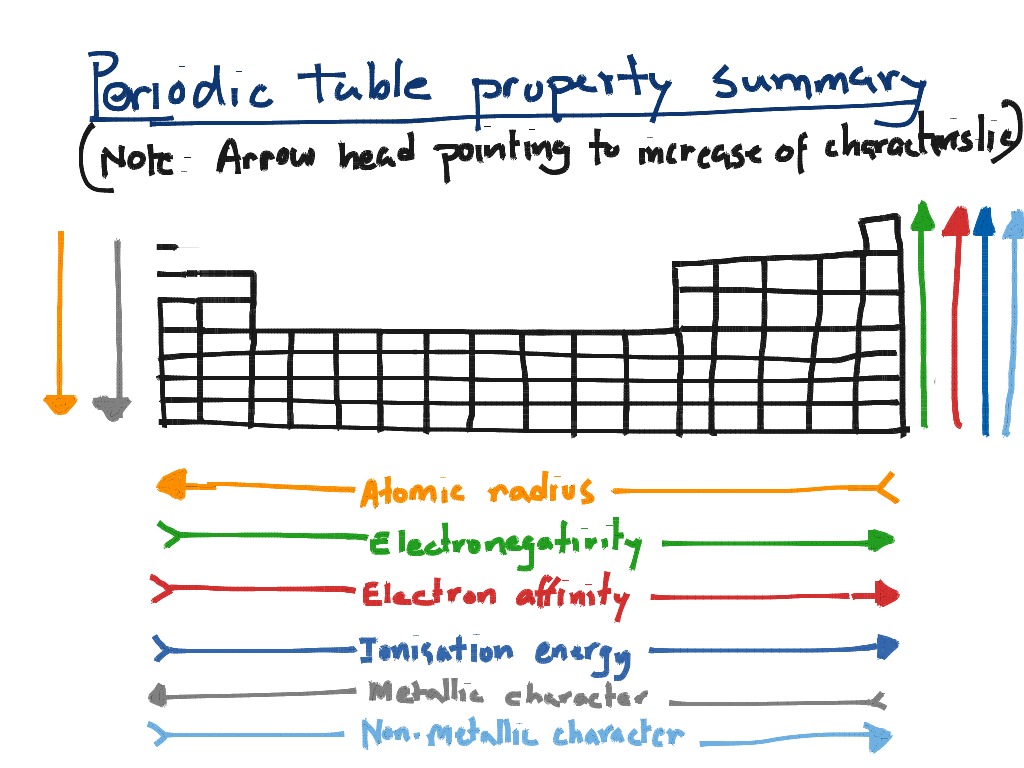
Refer to the arrangement of elements in the periodic table the horizontal row of element is called period and the same in vertical column is called group. In fact, this tabular representation of elements is the base for chemistry subject. 
In chemistry, understanding the basic periodic table facts is like learning the A, B, C… of the English language.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
